Esch, M.B.; Sukhorukov, V.L.; Kürschner, M.; Zimmermann, U. “Dielectric Properties of Alginate Beads and Bound Water Relaxation Studied by Electrorotation” Biopolymers, 1999, 50, 227-237
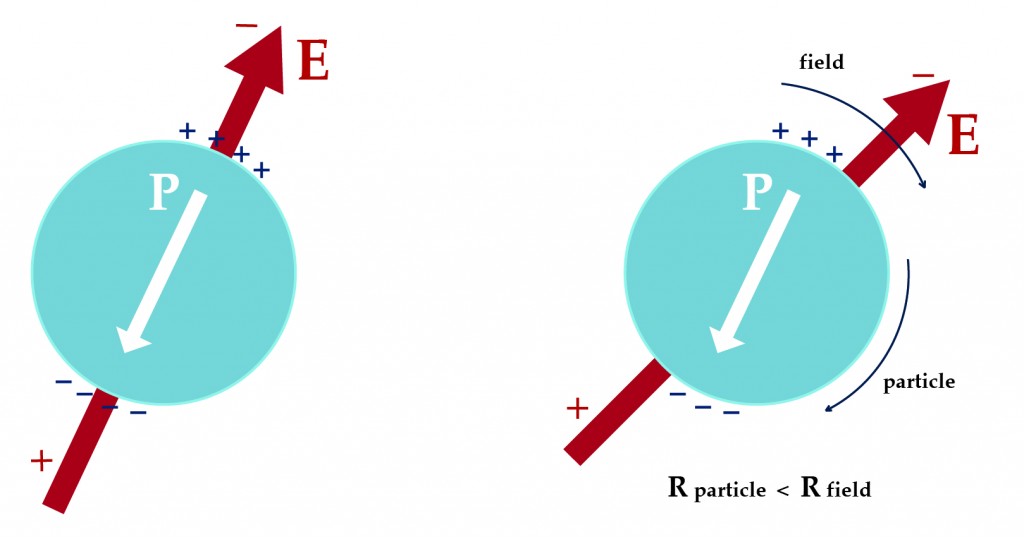
The association of water with polysaccharides is the basis for the survival of cells within hydrogels. Hydrogel-encapsulated cells are important because they could replace native cells that have experienced a loss of function, for example insensitivity to insulin-needs of the body. Here we proved experimentally that water is bound non-covalently to hydrogels. To prove this, we used four microelectrodes to create a rotating electric field in which we placed 400 micrometer large alginate beads. We measured how fast the beads rotated in fields of varying frequency and in medium of varying conductivity. A broad internal dispersion of the hydrogel centered between 20 and 40 MHz. We attribute this dispersion to the relaxation of water bound to the polysaccharide matrix of the beads.