Mandy B. Esch, Jean Prot, Ying Wang, Paula Miller, Micheal L. Shuler
Introduction: Toxicity to the liver is one of the most common reasons for drug attrition during clinical trials. Liver cell culture devices that increase the sensitivity of liver cells to drug actions could help in identifying drugs that are toxic more easily. To decrease the risk posed to humans and animals, tests with in vitro tissues precede trials with animals, and tests with animals precede clinical trials with humans. Despite extensive testing, one cannot always predict drug toxicity accurately because animals and in vitro tissues do not recapitulate human tissues and metabolism as accurately as we need it. In this presentation we will discuss a liver cell culture device that could help improve early stage drug testing and also contribute to clarifying the mechanisms that enable hepatocytes to increase their metabolism in response to fluidic cell culture conditions.
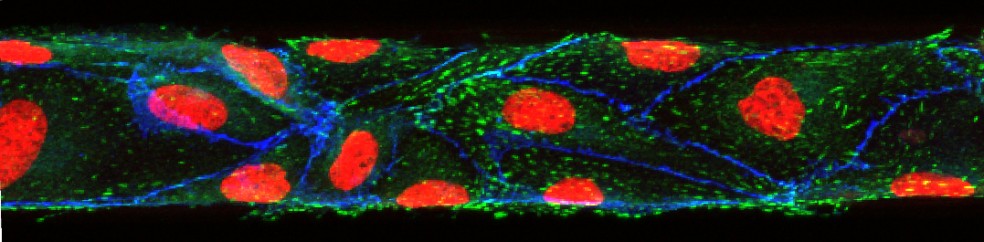
Results: To create the fluidic flow in our devices in an inexpensive manner, we used a rocking platform that tilts the device at angles of ±12°, resulting in a periodically changing hydrostatic pressure drop and bidirectional fluidic flow (average flow rate of 650 µL min-1, and a maximum shear stress of 0.64 dyn min-2). We tested the performance of this cell culture device by co-culturing human primary non-parenchymal cells (fibroblasts, stellate, and Kupffer cells) with human primary hepatocytes for 14 days, finding that hepatocytes produced albumin and urea at elevated levels compared to static cultures. This result confirms our hypothesis that, similar to unidirectional flow, periodically changing bidirectional flow enhances the metabolic activity of hepatocytes. Hepatocytes also responded with P450 (CYP1A1 and CYP3A4) enzyme activity when challenged with P450 inducers throughout the 21 days of device operation. Non-parenchymal cells were similarly responsive, producing interleukin 8 (IL-8) when challenged with 10 µM bacterial lipoprotein (LPS). Flow rates were passively controlled via the dimensions of the microfluidic channels. Our results indicate that device operation with bi-directoinal gravity-driven medium flow supports the long-term culture of primary human liver cells with the benefits of enhanced metabolic activity. Our mode of device operation allows us to evaluate drugs under fluidic cell culture conditions and at low device manufacturing and operation costs.
 Mahler, G.J., Esch M.B., Shuler, M.L. “Characterization of a Gastrointestinal Tract Microscale Cell Culture Analog Used to Predict Drug Toxicity ”, Biotechnology & Bioengineering, 2009, 104(1), 193-205.
Mahler, G.J., Esch M.B., Shuler, M.L. “Characterization of a Gastrointestinal Tract Microscale Cell Culture Analog Used to Predict Drug Toxicity ”, Biotechnology & Bioengineering, 2009, 104(1), 193-205.